 
Several commercial firms have offered to aid in scaled-up production, but the inherent variability of the manufacturing process and the risk of failure when scaling up so rapidly to such a high volume could create problems. 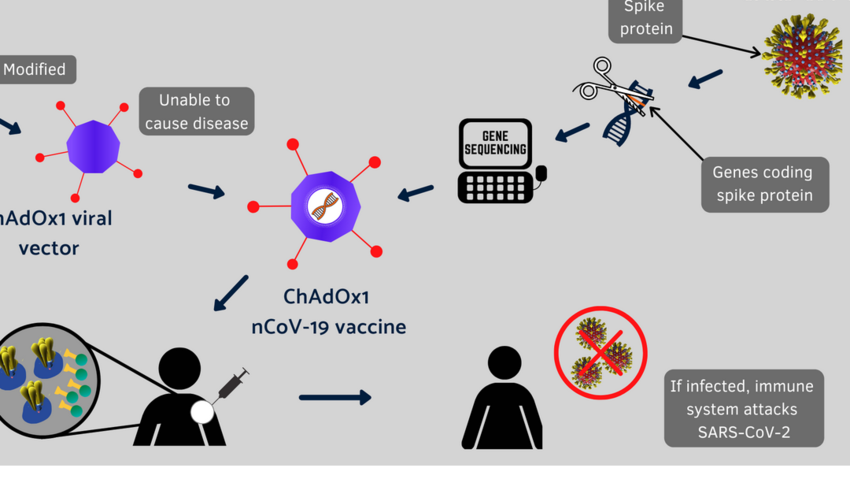
Currently, there is only one manufacturer of licensed anthrax vaccine, but production is limited because of regulatory problems. 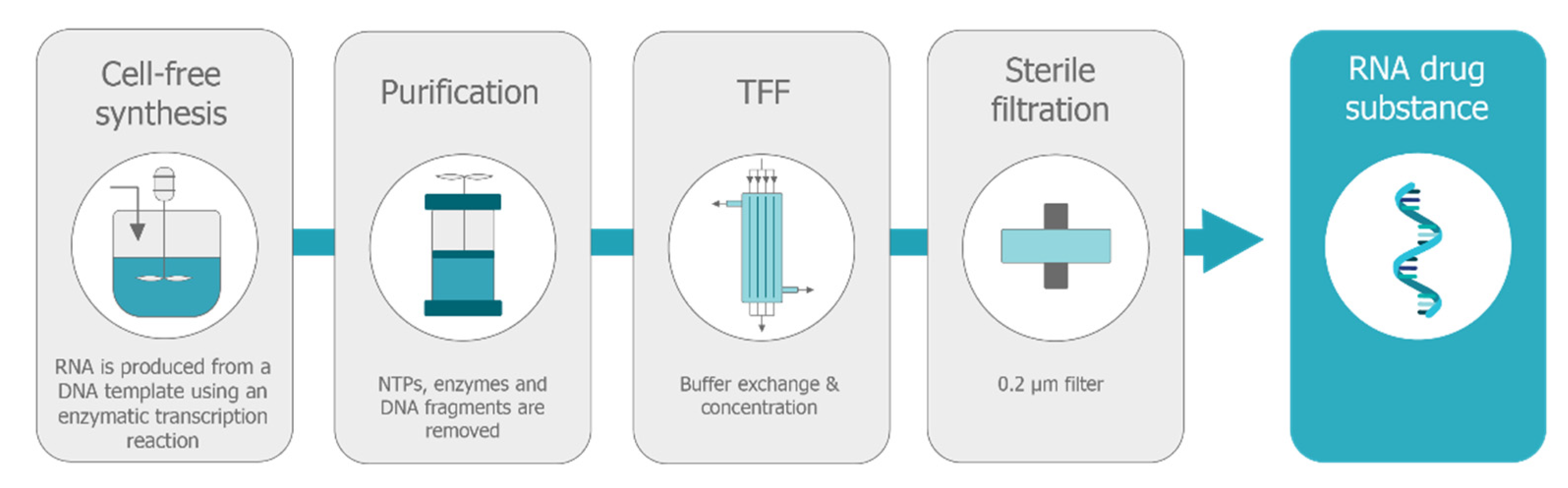
But there is an urgent need for more anthrax vaccine for the immunization of high risk civilian populations, as well as for use in medical management of exposed individuals in conjunction with antibiotics. As of May 2001, over two million doses of the current anthrax vaccine have been administered to over 500,000 individuals, mostly military personnel. Long-term goals include developing a safer vaccine that can be used in immunocompromised or other at-risk individuals.Īnthrax vaccine is another top priority. The vaccine immune globulin (VIG) supply also needs to be expanded. An aggressive clinical development plan is currently in place its goal is to build the stockpile with enough vaccine to protect the entire country within the year. The threat of a global pandemic makes smallpox one of the top vaccine priorities. Currently, there is no policy in place for immunizing the civilian population as a bioweapons defense measure, however several government agencies are working at unprecedented speed to put the correct policies into place. Additionally, DoD has developed vaccines to be used in normal healthy adults between the ages of 18 and 65, not pediatric, geriatric, immunocompromised or other subsets of the civilian population. The very nature of terrorism produces a high level of uncertainty about what to expect and how to prepare. Much of the challenge is due to the fact that the threats are uncertain and risk-benefit information difficult to assess. The use of vaccines as a civilian biodefense measure presents multiple challenges that are quite different from those of vaccine use by the military. It was suggested during this session that in order to move animal and clinical testing forward, incentives need to be established to reduce the current challenges of vaccine development vaccine production priorities need to be set and a central office or leader authorized to declare top priorities and the role of the major vaccine manufacturers needs to be facilitated by clear directions and active collaboration between industry and government. This is alarming considering the eight to ten years often needed to develop a new vaccine, compared to only two to three years to develop a new bioweapon.Įven among the four major vaccine manufacturers, there is insufficient production capacity. But the potential market has been too small and uncertain to encourage the vaccine industry to make large investments in research, development, and manufacturing of new products. From a bioterrorist perspective, vaccine-resistant agents are more difficult to engineer than drug-resistant agents. Vaccines not only afford the best protection against infectious disease but can serve as strong deterrence factors as well. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
